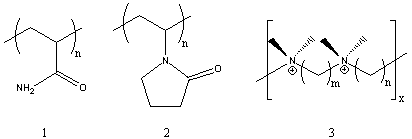
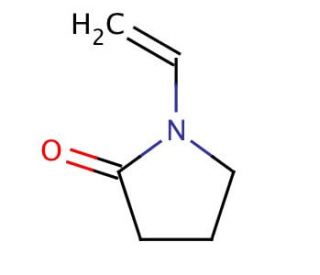
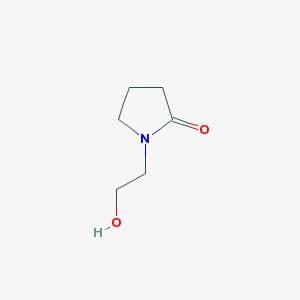
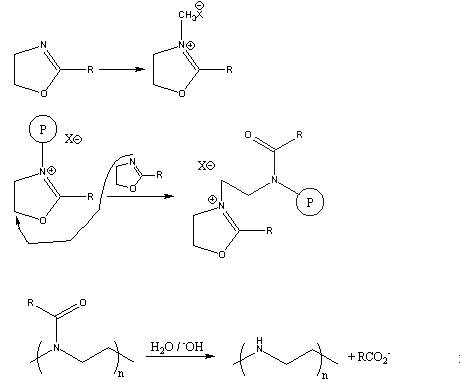
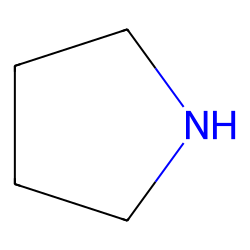
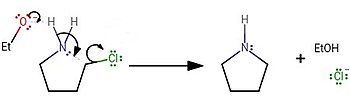
A Mild and Efficient Procedure for Ring-Opening Reactions of Piperidine and Pyrrolidine Derivatives by Single Electron Transfer Photooxidation - ScienceDirect
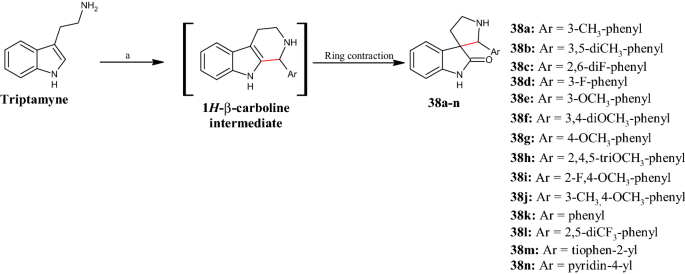
Pyrrolidine in Drug Discovery: A Versatile Scaffold for Novel Biologically Active Compounds | SpringerLink

Gold(I)-Catalyzed Domino Ring-Opening Ring-Closing Hydroamination of Methylenecyclopropanes (MCPs) with Sulfonamides: Facile Preparation of Pyrrolidine Derivatives

Removal of various anionic dyes using sodium alginate/poly(N-vinyl-2- pyrrolidone) blend hydrogel beads | Semantic Scholar
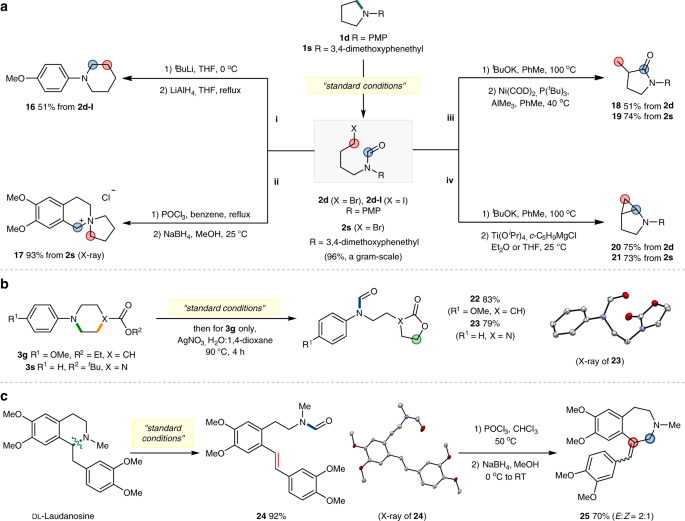
Ring-opening functionalizations of unstrained cyclic amines enabled by difluorocarbene transfer | Nature Communications

2-Pyrrolidinones and 3-Pyrrolin-2-ones: A Study on the Chemical Reactivity of These Structural Moieties

Synthesis of eight-membered rings and a pyrrolidone ring-fused compound. | Download Scientific Diagram

Significance of pyrrolidone ring in the precipitation-based reprocessing for spent nuclear fuels | WRHI – Tokyo Tech World Research Hub Initiative

2-Pyrrolidinones and 3-Pyrrolin-2-ones: A Study on the Chemical Reactivity of These Structural Moieties

Synthesis of biobased N-substituted pyrrolidone ring-based polyamides... | Download Scientific Diagram
A curious case of dynamic disorder in pyrrolidine rings elucidated by NMR crystallography - Chemical Communications (RSC Publishing)
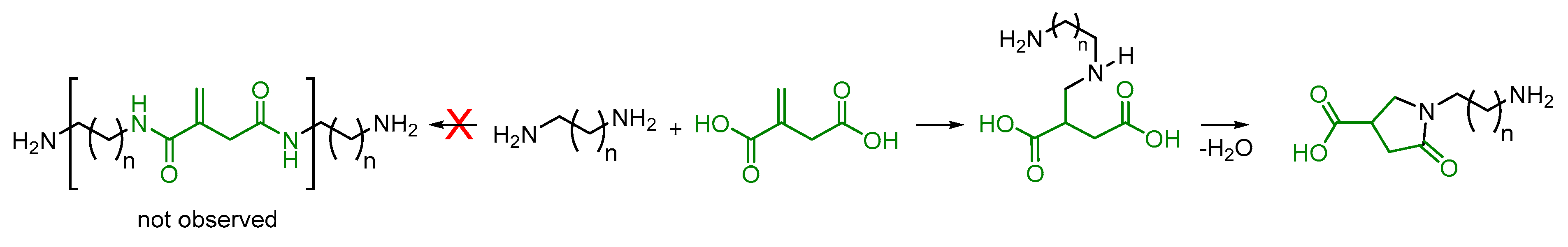
Applied Sciences | Free Full-Text | First Example of Unsaturated Poly(Ester Amide)s Derived From Itaconic Acid and Their Application as Bio-Based UV-Curing Polymers | HTML
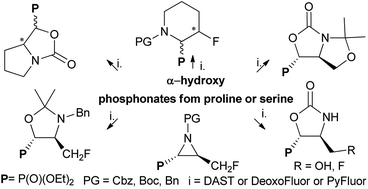
Pyrrolidine and oxazolidine ring transformations in proline and serine derivatives of α-hydroxyphosphonates induced by deoxyfluorinating reagents - RSC Advances (RSC Publishing)